Current projects
Consortia in which our projects are embedded: SFB-TR84, NUM-Organo-Strat, RAPID, EC3R.
Our research is dedicated to the inflammatory and immunological analysis of the cellular and molecular host-pathogen interaction in the human alveolar compartment. By use of our central experimental model of living human lung tissue as well as lung organoids, we simulate the onset of pneumonia by infection with important pathogens such as Streptococcus pneumoniae, influenza A virus, (SARS-)corona viruses, Staphylococcus aureus, or e.g. Legionella pneumophila.
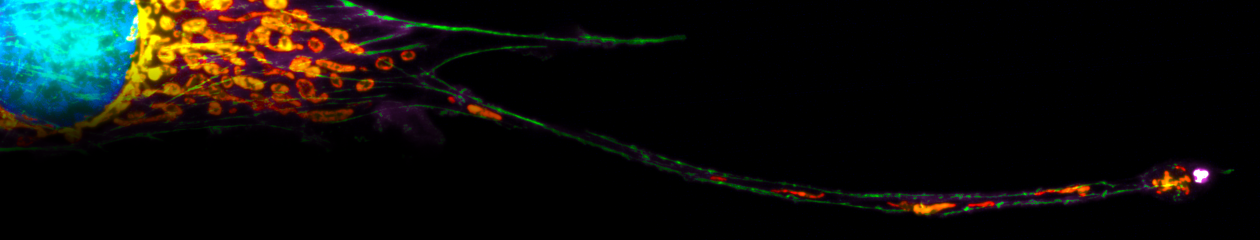
A broad spectrum of molecular biological approaches and biochemical assays help us to analyze the molecular communication between e.g. alveolar macrophages and alveolar epi- and endothelial cells. We are following these processes especially with high-end fluorescence microscopy to visualize infection live and in color.
High-end microscopy in the cellular microbiology of the lung is our passion. We are pleased to contribute with these techniques as one of the two core projects to the SFB-TR84, „Innate Immunity of the Lung – Mechanisms of Pathogen Attack and Host Defense in Pneumonia“. By this, we support many sub-projects of the SFB in the direction of molecular imaging in the pulmonary pathogen-host interaction. Various microscopic techniques are used: life-cell and -tissue imaging, FLIM-FRET, FCS, FRAP, spectral imaging and linear unmixing, or super-resolution microscopy.
Our projects are currently focussing e.g. on the cellular tropism / immune activation of SARS-/MERS-CoV and its related virulence. Other projects aim at elucidating the role of mitochondrial dysfunction and its contribution to cell death and innate immune activation after pneumococcal or influenza infection.
Next to these projects, we are taking strong efforts to develop new techniques for 3D based human disease models. These include new cutting techniques based on high-pressure water jet for native tissue, adenoviral transduction with CRISPR/Cas, cryo-conservation of human lungs and organoids for biobanking, AI-based image analysis etc. This work has two major reasons. The first is to find new robust and reliable alternative models for bio-medical research to existing animal models, which often do not reflect the (immunological) aspects of human disease and are therefore often misleading for the validation of new therapeutic agents. The second is the ethical reason to slowly overcome the philosophical view of the „Speziesismus„, which is the moral discrimination of beings on the base of belonging to a certain species, or – being animal and not human. The model we established was awarded with the first research prize for Animal Experimental Alternatives of the State of Berlin in 2011.